Three completed studies have produced a consistent profile supporting taplucainium’s development in rCC/uCC. Across preclinical, Phase 1, Phase 2a, and bridging studies, taplucainium has demonstrated rapid onset of cough reduction and a favorable tolerability profile, supporting the ongoing Phase 2b ASPIRE study, which is designed to evaluate once-daily taplucainium inhalation powder in adults with rCC/uCC.
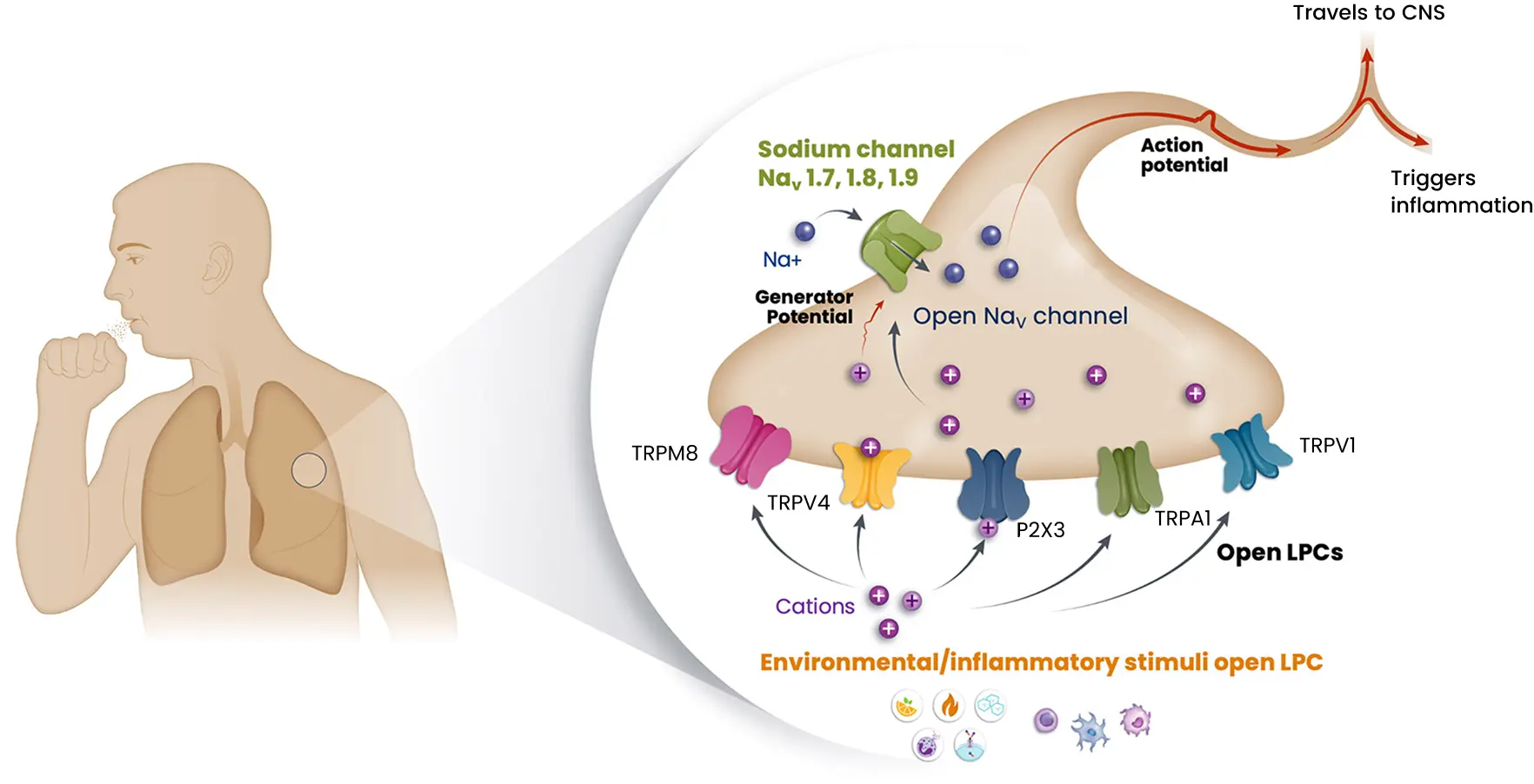
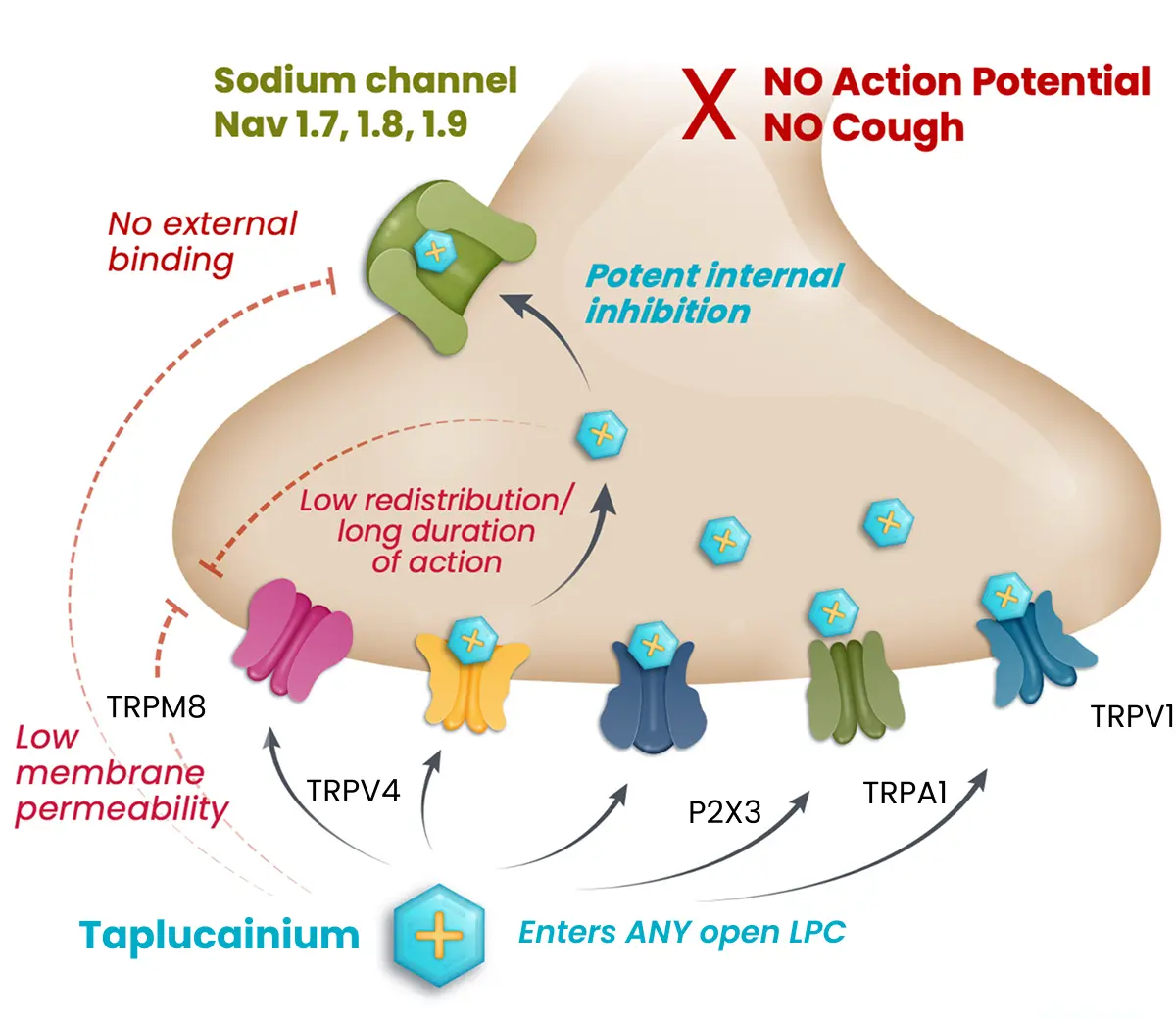
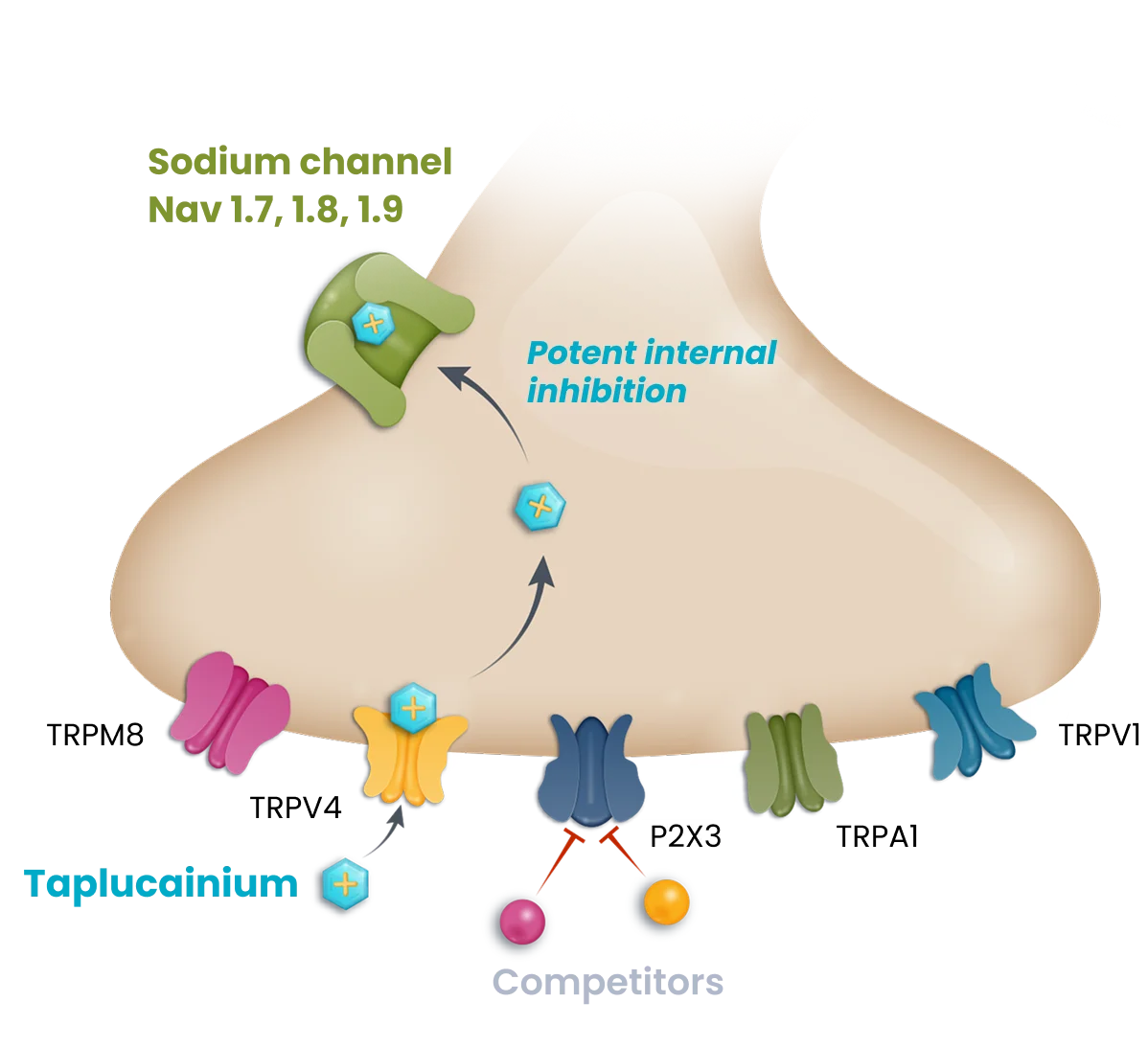
With delivery directly to the airway by a dry powder inhaler, taplucainium enters nociceptors through open large pore channels and blocks the signaling to the central nervous system that leads to the cough response. This unique mechanism is intended to provide fast-acting and durable cough control with a profile designed to avoid limitations that have constrained prior investigational approaches in chronic cough. Follow the steps below to learn more about how taplucainium works.